NEJM Reports: Substandard Generic Drugs — Threats to Patient Safety and National Security
Mar 18, 2026

By Kevin Schulman, M.D. & Arthur L. Kellermann, M.D., March 18, 2026 | From: NEJM
Generic drugs account for more than 90% of prescriptions filled in the United States. The first paragraph on the home page of the Office of Generic Drugs at the Food and Drug Administration (FDA) asserts that “FDA-approved generic drugs have the same high quality, strength, purity and stability as brand-name drugs.” On the strength of this assurance, America’s doctors, pharmacists, and patients assume that every version of a generic drug is equally safe. But this proposition is now being seriously challenged.
Back in 1984, the Hatch–Waxman Act established an “abbreviated new drug approval” process for generic drugs. This new pathway led to a sharp drop in drug prices. Between 2009 and 2019, the availability of generic medicines saved U.S. patients $2.2 trillion, according to the FDA.
Over time, intense price competition drove most production of generic drugs and ingredients offshore to countries with low labor costs and lax regulatory controls. Once that shift occurred, relentless pressure to minimize costs led some manufacturers to compromise on quality. Rapid globalization also outstripped the FDA’s capacity to monitor manufacturers. In 2022, the Government Accountability Office reported that 61% of foreign plants had not been inspected by the FDA in the preceding 5 years.1
When FDA inspectors finally reach these plants, some find glaring problems. In 2023, inspectors in India reported that Intas Pharmaceuticals employees had poured acid over quality-assessment documents to keep them from being reviewed. In 2025, an FDA report noted that employees at a Hetero Pharmaceuticals plant destroyed manufacturing documents and stored drug ingredients in an unregistered warehouse where inspectors found birds, lizards, and cats.
More than 60% of generic-drug shortages are attributable to quality concerns, according to the FDA. Shortages are most likely to happen when production is concentrated in a few plants and problems with quality are found at one of them. For example, the Intas Pharmaceuticals plant that failed its 2023 inspection was one of only two suppliers of cisplatin, a vital cancer drug, to the United States. The failure triggered a months-long shortage.
In another instance, a private-sector laboratory detected high levels of nitrosamines (known carcinogens) in drugs made by several FDA-approved manufacturers, prompting recalls of metformin, angiotensin-receptor blockers, angiotensin-converting–enzyme inhibitors, prazosin, and ranitidine. More recently, independent tests of generic methylphenidate found nitrosamine levels above the FDA’s safety threshold in 7 of 15 immediate-release products.2
Low-quality drugs can harm patients. Although there was not much interest in this issue when generic drugs were assumed to be high quality, cases of organ failure have been linked to poor-quality immunosuppressive drugs.3 Recently, a team of U.S. and South Korean researchers with access to FDA data determined that significantly more serious adverse event reports were linked to generic drugs manufactured in India than to equivalent drugs manufactured in the United States.4
The fragility of America’s global supply chains complicates FDA enforcement. In 2008, a total of 238 deaths in the United States were linked to adulterated Chinese heparin. When the FDA toughened its approach to quality assessment of foreign manufacturers, shortages of more than 200 medications followed. This crisis prompted the FDA to prioritize minimizing drug shortages over ensuring safety. In some instances, overseas companies were allowed to ship shortage drugs to the United States from plants the agency had banned from exporting drugs to the United States. Congress, doctors, and patients were not informed, according to ProPublica.
There is a better way to assure the safety of generic drugs. In 1994, the European Medicines Agency (EMA), for example, established a proactive approach involving risk-based surveillance in addition to systematic planned and ad hoc testing of generic drugs both on the market and during routine inspections of manufacturers (in contrast, the FDA does not routinely test generic-drug products themselves, either on the market or during quality inspections of manufacturing plants). EMA testing relies on a network of official medicines control laboratories (OMCLs) that operate in accordance with International Organization for Standardization (ISO) accreditation standards for testing and calibration laboratories. At any point in a drug’s life cycle, an OMCL can pull samples for product testing. If they couldn’t do so, an OMCL brochure asserts, “patients and users of medicines in Europe could be exposed more often to defective (e.g., contaminated) or falsified and illegal products.”
Five actions could leverage market forces to ensure drug safety in the United States. The first is acknowledging the problem. The FDA should stop claiming that all generic drugs sold in the United States are equally safe and effective. It cannot verify this claim without product testing. Moreover, recent budget and personnel cuts may make the agency’s oversight of generic-drug quality even more challenging.
Second, to compensate, the FDA can encourage testing of generic-drug quality by independent, ISO-accredited laboratories. Recently, a group of pharmaceutical experts developed a scoring system that translates test results and past regulatory data into a readily interpretable rating scale.5 Data from an ongoing evaluation of essential medicines for the U.S. Department of Defense illustrate the value of this approach (see table). The FDA could use test results to prioritize its factory inspections and strengthen oversight.
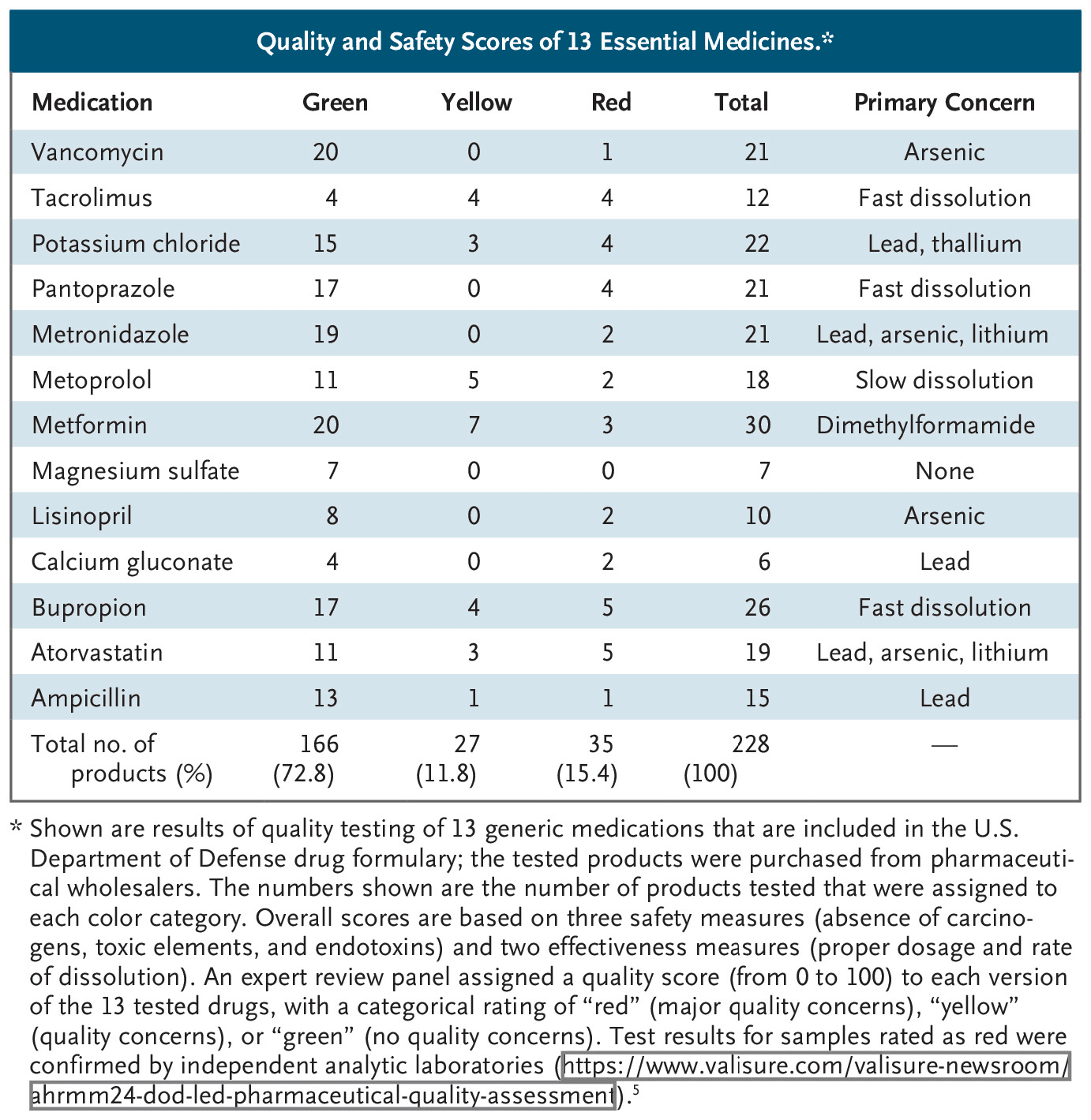
Quality and Safety Scores of 13 Essential Medicines.
Third, federal agencies, including the Centers for Medicare and Medicaid Services, could purchase drugs on the basis of best value rather than lowest cost. High-quality generic drugs are often available at prices similar to those of low-quality products. Consistent use of only high-quality generic medications could reduce costs by preventing complications and improving outcomes.
Fourth, quality scores could be made transparent to consumers. Kaiser Permanente does not buy certain generic medicines unless they are independently certified by a laboratory of its choice. If drug-quality information were widely available, patients, health systems, and pharmacies would reward high-quality suppliers with contracts and market share. Structurally, transparency would drive the market toward higher quality more quickly and definitively than episodic FDA plant inspections can do.
And fifth, the U.S. government should oversee an effort to rebuild America’s capacity to manufacture generic drugs, combining investment in private manufacturing with incentives for purchasing U.S.-made products under the Medicare and Medicaid programs. Currently, the United States is vulnerable to an embargo of essential drugs or the materials required to make them. A recent evaluation for the Department of Health and Human Services found that 87% of sites that make active pharmaceutical ingredients (APIs) and 63% of sites that produce finished dosage forms were located overseas. In 2021, a private holding company bought the only U.S. manufacturing plant licensed for the production of amoxicillin out of bankruptcy. Bringing generics manufacturing back to the United States is in our national interest. Advanced pharmaceutical manufacturing, pioneered by the Medicines for All Institute in Virginia, produces high-quality APIs at lower cost than traditional factory methods. CivicaRx, a not-for-profit generics company, supplies member hospitals by paying high-quality manufacturers a sustainable price under long-term contracts. We believe that both approaches should be more widely embraced.
Most generic drugs are safe, but a troubling minority are not. Testing finished drugs, signing “green” manufacturers to long-term contracts, and sharing quality scores with the public would help realign market forces to improve safety. The United States already tests a wide range of consumer products. We should also test our generic drugs.


















